Table of Contents

Hiroshi Miyake (hereinafter “Miyake”): My name is Hiroshi Miyake and I’m Representative Director of Chordia Therapeutics. Thank you for joining us today. I’d like to present our financial results for the fiscal year ended August 31, 2025.
Today’s presentation will follow the agenda shown on the slide.
Chordia is a Small-Molecule Anti-Cancer Drug Discovery Company

We were founded in October 2017 as a spin-off from Takeda Pharmaceutical Company Limited, Japan’s largest pharmaceutical company. We are a biotech startup engaged in the research and development of small-molecule anti-cancer drugs. In June 2024, we were listed on the Tokyo Stock Exchange Growth Market, under the securities code 190A.
Headquartered at the Shonan Health Innovation Park (commonly known as Shonan iPark) in Fujisawa City, Kanagawa Prefecture, we have 23 employees, including 12 with Ph.D. degrees, and operate as a highly specialized team dedicated to developing next-generation anti-cancer drugs. Our largest shareholder is Takeda Pharmaceutical Company Limited, our parent company at founding, which holds approximately 15% of our shares.
Chordia’s Business Model is Based on a High-value Pipeline with an Established Clinical POC
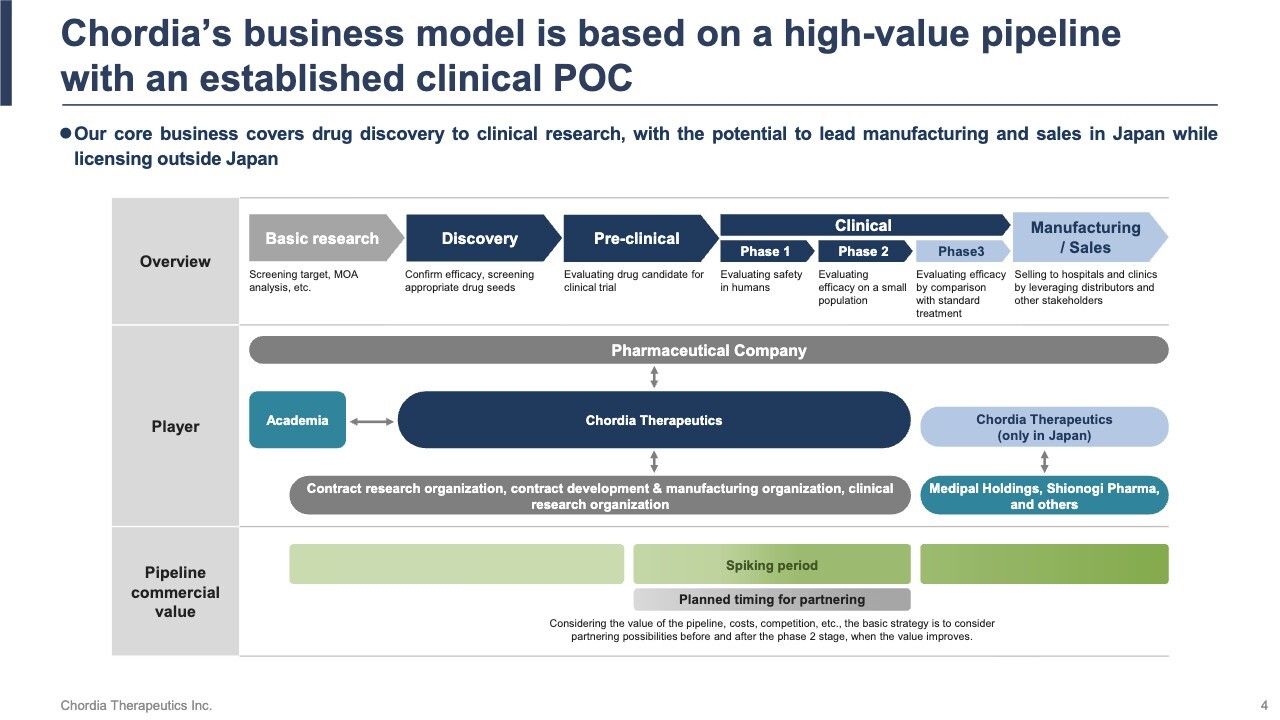
Developing a new drug typically takes around 20 years. The first 10 years focus on basic research at universities and other institutions, followed by discovery research and pre-clinical studies that lead to the start of human clinical trials. The next 10 years involve confirming safety and efficacy through clinical trials, securing regulatory approval and pricing, and finally reaching the market. This long journey is truly a marathon-like challenge.
Within this 20-year process, we have adopted a strategy to conduct all processes from discovery research through Phase 2 clinical trials in-house. This strategy is driven by a clear objective: to shorten development timelines and accelerate value creation.
Specifically, we outsource basic research to Japanese universities, integrating cutting-edge insights through collaborative research. Japan’s universities rank among the world’s best in science and technology—evidenced by this year’s Nobel Prize winners—and we leverage this strength to the fullest.
By focusing on rare cancers, we increase the likelihood of obtaining accelerated approval based on Phase 2 clinical trial results, allowing us to reach the market faster than usual. Furthermore, if favorable results are achieved in Phase 2 trials, the value of our pipeline will rise significantly, enabling out-licensing opportunities and early monetization.
While drug discovery is typically a high-risk, high-return business, we have established a framework that allows us to control risk and enhance our probability of success. More than half of our team members hold doctoral degrees, forming a small but highly skilled team with development capabilities on par with those of major pharmaceutical companies.
With both speed and flexibility in decision-making, we advance development swiftly while maintaining scientific excellence. Through this structure, we aim to achieve highly reliable value creation in what is inherently a high-risk, high-return business.
Balance Sheet as of Fiscal Year Ending August 2025
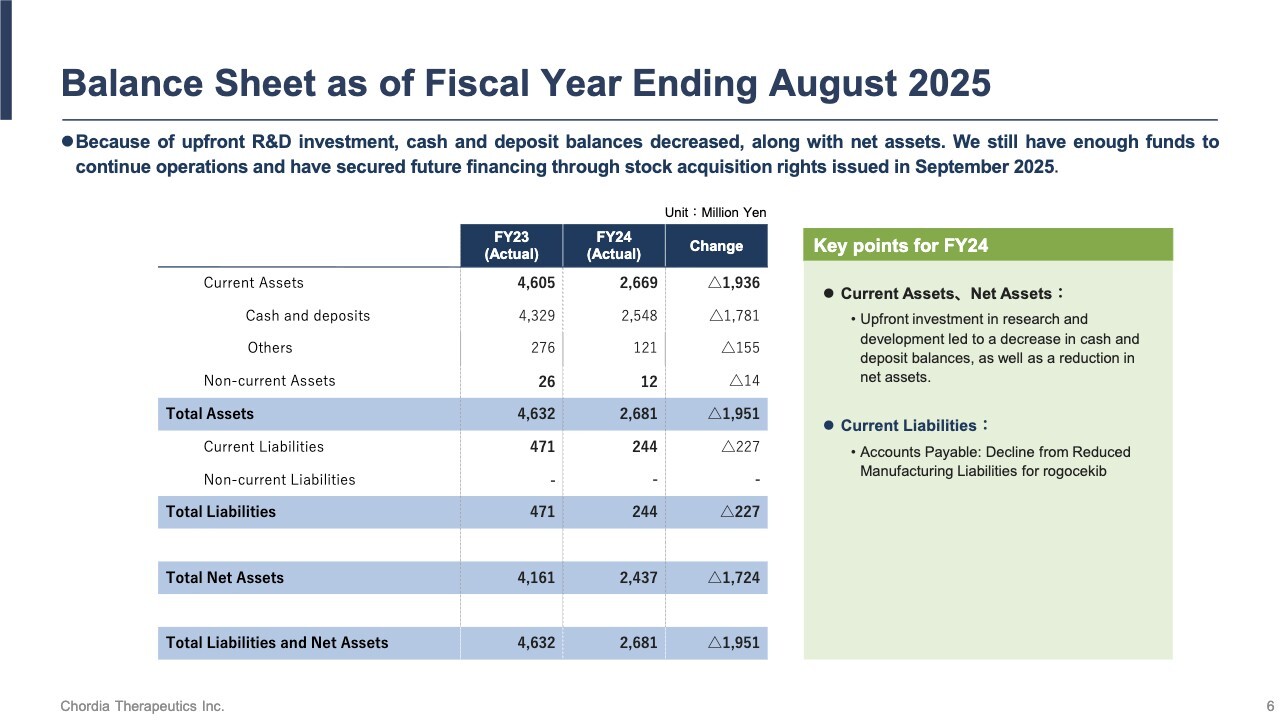
As of August 31, 2025, we held approximately ¥2.5 billion cash and deposits, ensuring the funds sufficient to sustain our operations. Additionally, we are advancing financing through stock acquisition rights to secure medium-term funding for the development of our lead pipeline, rogocekib.
Profit or Loss as of Fiscal Year Ending August 2025
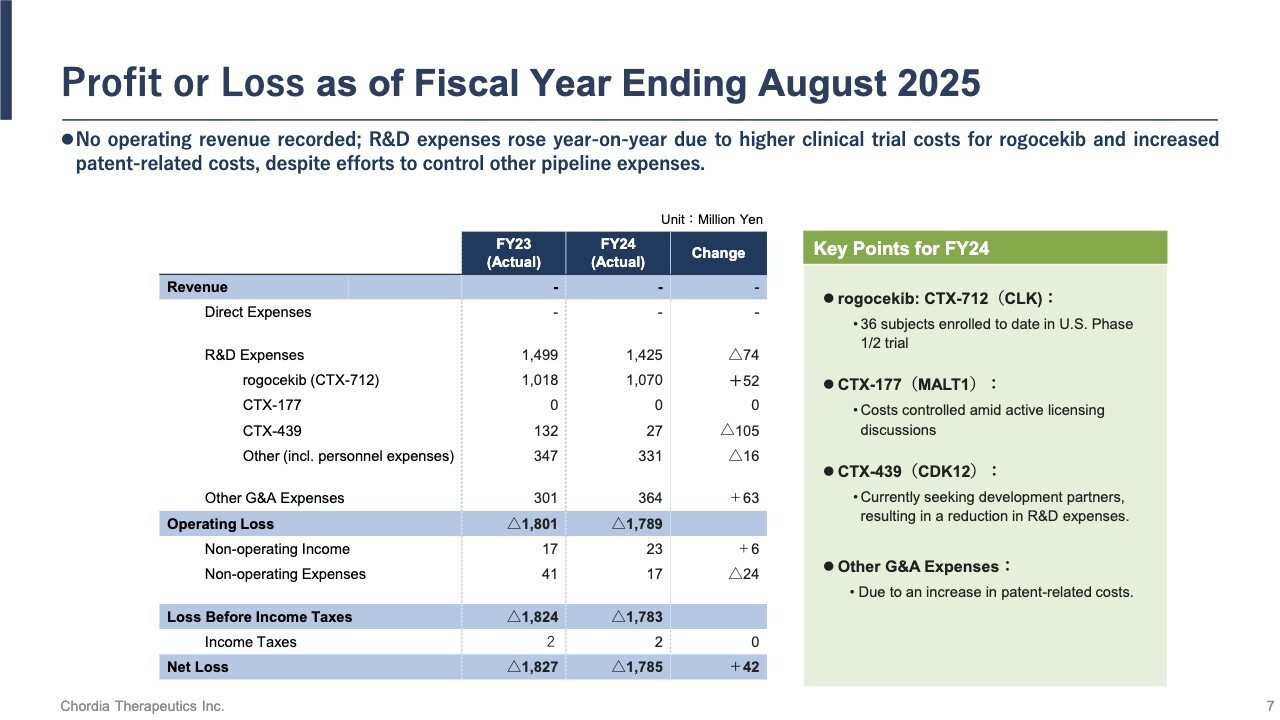
Let me summarize our operating results. For the fiscal year ended August 31, 2025, we recorded no revenue. R&D expenses totaled approximately ¥1.4 billion, of which roughly ¥1.0 billion was allocated to the development of rogocekib.
Patient enrollment in the U.S. clinical trial has progressed largely as planned, and as of the end of August, we completed dosing for 36 patients. As a result, we reported a net loss of ¥1,785 million.
Performance Forecast for Fiscal Year Ending August 31, 2026
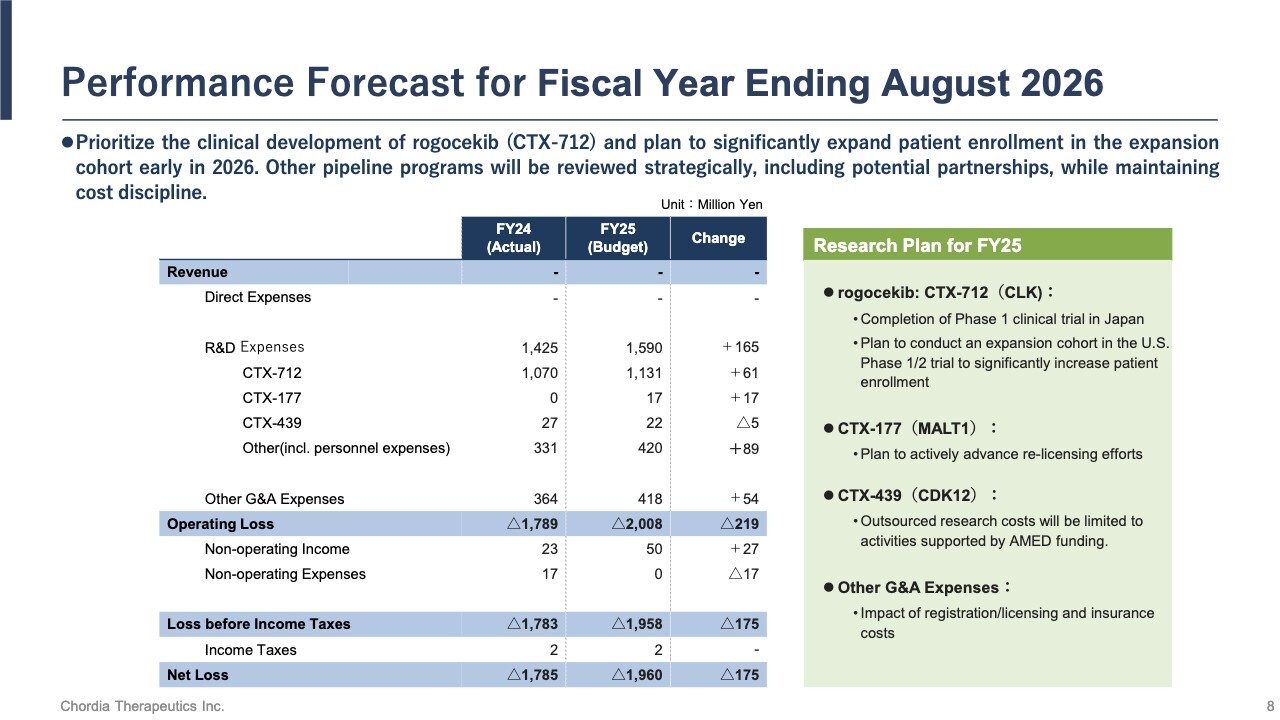
For the fiscal year ending August 31, 2026, we will continue to prioritize the development of rogocekib. We plan to launch an expanded cohort early in 2026 to significantly increase patient enrollment. For our other pipeline programs, we will manage costs carefully while strategically exploring partnerships with other companies.
Based on the above, we plan to allocate ¥1,590 million to R&D expenses and project a net loss of ¥1,960 million.
RNA Generation Process and Pipeline Modes of Action
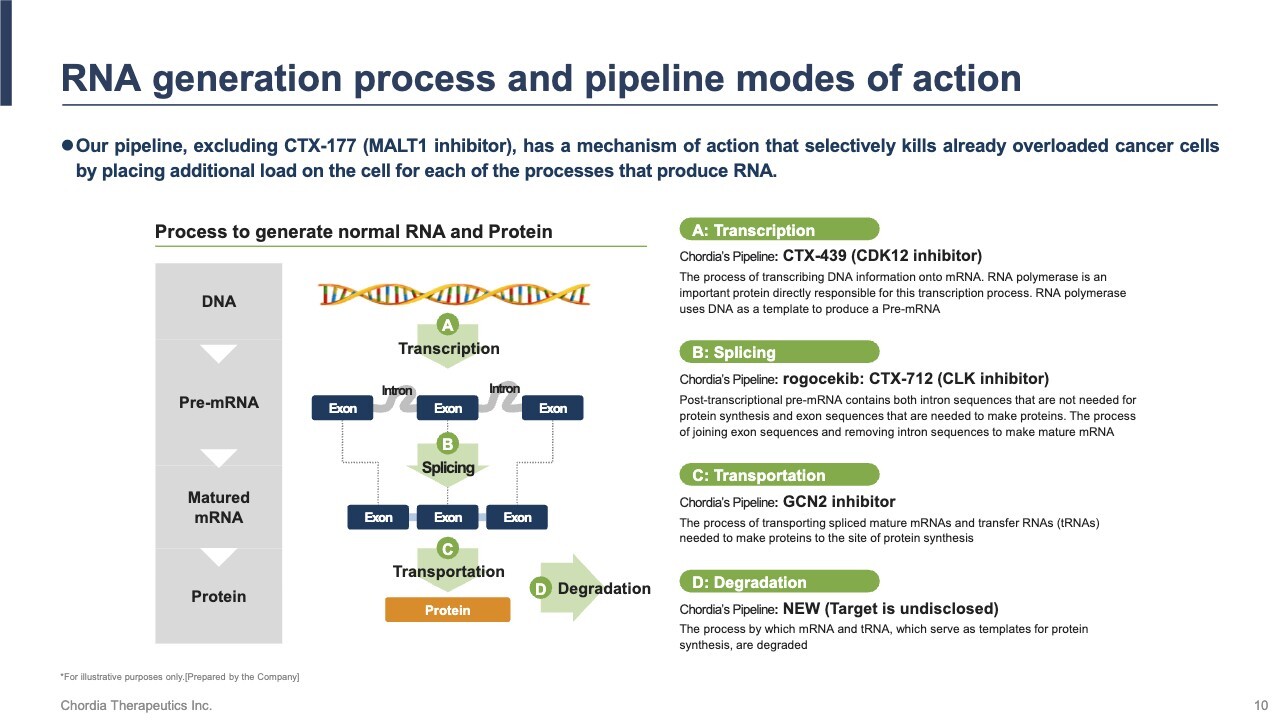
Let me update you on our pipeline progress. We focus on exploiting RNA deregulation stress, a vulnerability in cancer cells. RNA deregulation stress occurs when errors arise during RNA production, placing a burden on the cell. As shown on the slide, this process involves transcription, splicing, transportation, and degradation. In cancer cells, these processes are disrupted, leading to accumulated stress. Our pipeline is designed to amplify this stress further, selectively killing cancer cells.
RNA deregulation stress was discovered 14 years ago in 2011 by our scientific advisor, Professor Seiji Ogawa at Kyoto University. We initiated clinical trials in 2018, and they are now in their seventh year. We aim to submit for regulatory approval within the next three years, entering the final stage of nearly 20 years of research and development.
Concept of Stressing Out Cancer to the Point of No Return

The slide illustrates the level of stress experienced by cells using the size of balloons as a metaphor. Our pipeline selectively kills cancer cells by increasing stress, while normal cells remain largely unaffected due to their inherent stress tolerance.
Cancer cells already carry high stress, represented by fully inflated balloons. Normal cells, with low stress, have small balloons. When our pipeline applies additional stress, normal cells manage it, but cancer cells cannot cope—their balloons “burst,” leading to selective cell death.
This concept has been clinically validated by Takeda’s blockbuster anti-cancer drug, Velcade. We aim to achieve similar success using a novel mechanism: RNA deregulation stress.
Of the Five Pipelines, Two are in the Clinical Stage – Strategic focus on rogocekib, which has high market potential, to accelerate value creation
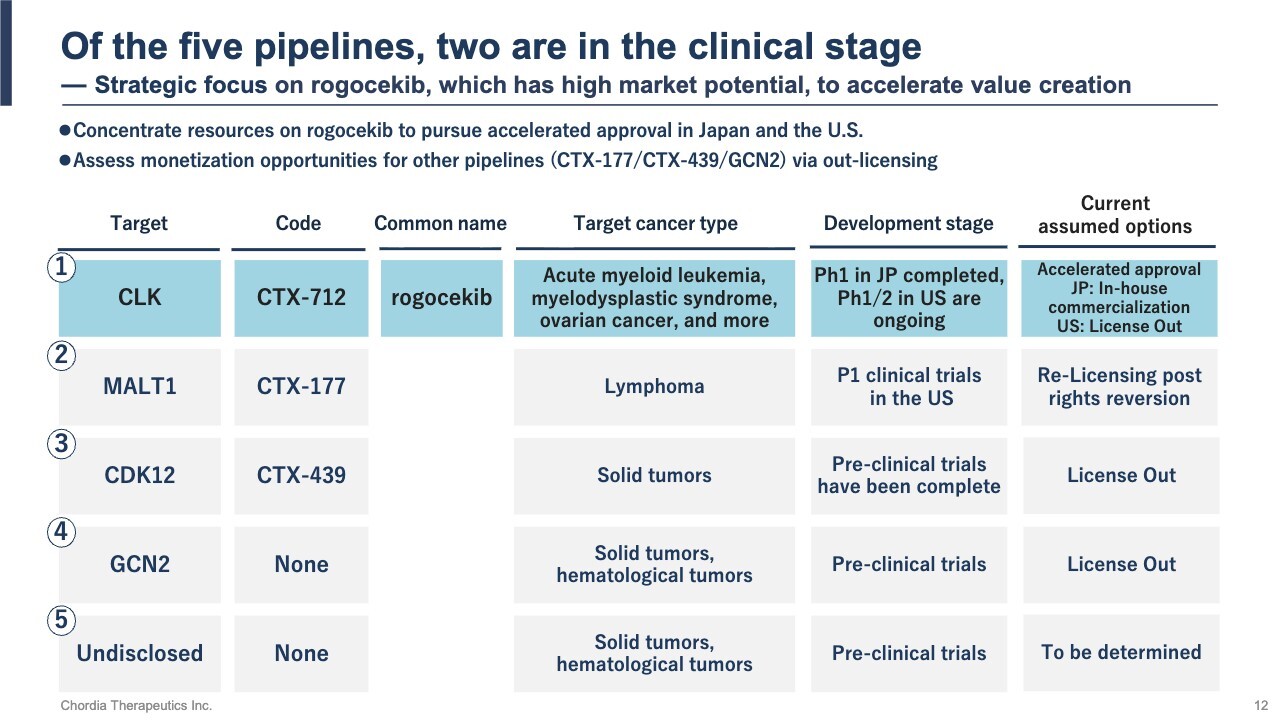
This slide shows our pipeline. We have five small-molecule pipelines, two of which —rogocekib and CTX-177—are currently in clinical trials.
We are concentrating our development resources on rogocekib, which has high market potential and the possibility of accelerated approval. For the other pipelines, we are pursuing value creation while controlling costs, exploring partnerships or licensing opportunities.
We aim to manage risk while securing multiple revenue opportunities through this focused and diversified approach.
rogocekib: An Innovative Small-Molecule Therapy for AML – Addressing JPY200 Billion + Market Growing at Over 10% Annually
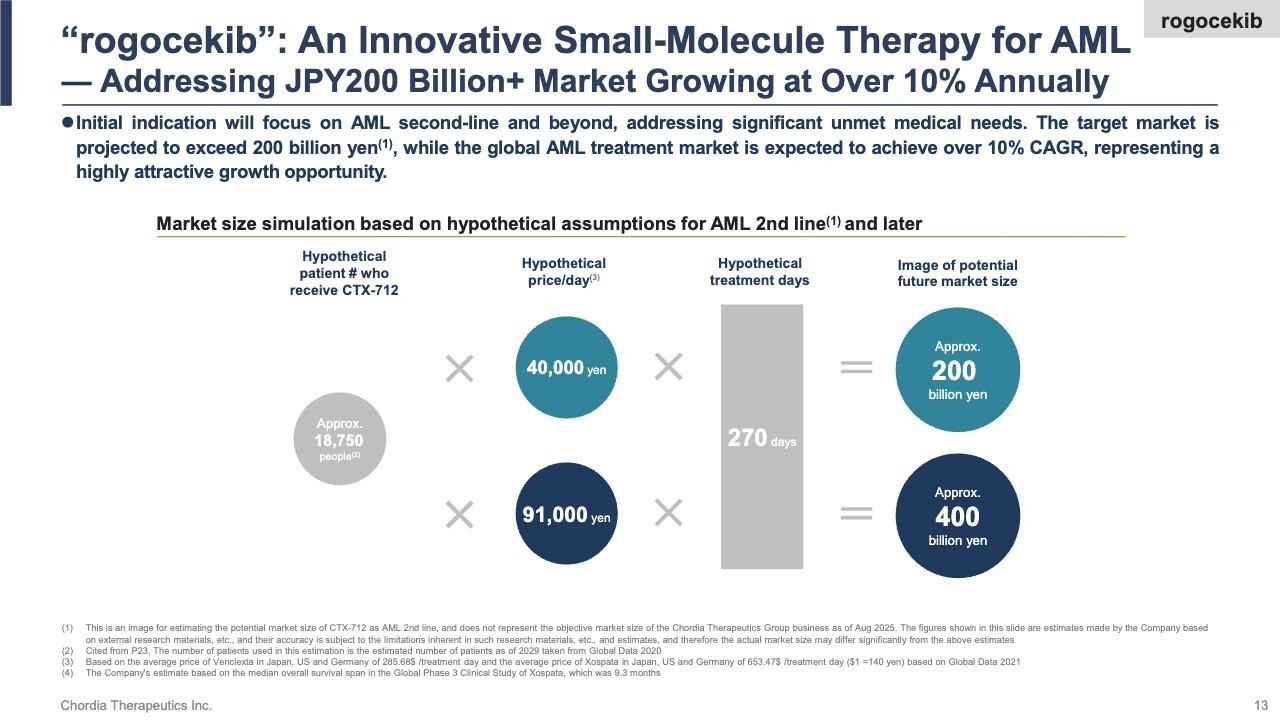
We are advancing clinical trials of rogocekib, aiming for the first approval in relapsed or refractory acute myeloid leukemia (AML). The slide illustrates its market potential.
We estimate that roughly 18,000 AML patients annually in Japan, the U.S., and Europe—who fail first-line therapy and require second-line therapy, and are ineligible for existing anti-cancer drugs—could benefit from rogocekib. Assuming an average treatment duration of nine months and a daily price of ¥40,000–¥90,000, based on comparable AML therapies, we see a potential annual market exceeding ¥200 billion.
The global AML treatment market is projected to grow over 10% annually, making it a highly promising and expanding sector. While rogocekib will not capture the entire market, securing just half of it could drive peak annual sales above ¥100 billion, positioning the drug as a potential blockbuster drug.
rogocekib: Promising Phase 1 Results in Japan – Demonstrating Competitiveness Against Recently FDA-Approved Therapies
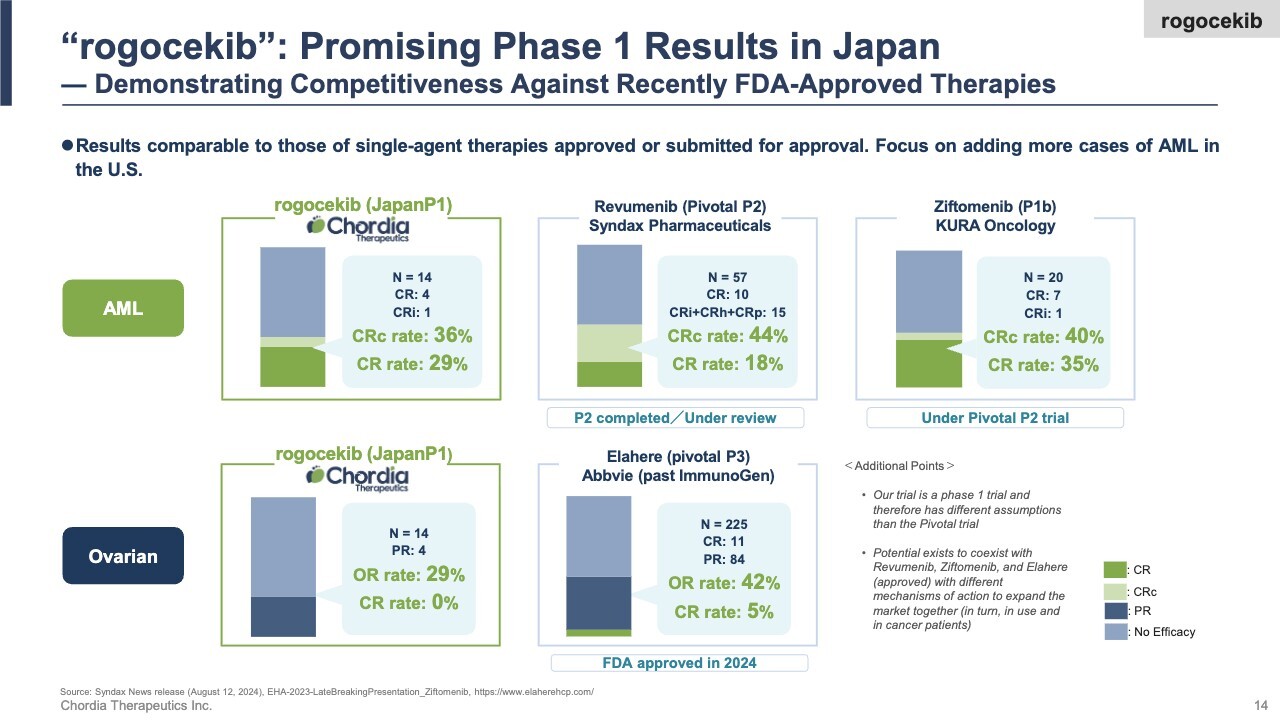
Here are the Phase 1 clinical results for rogocekib conducted in Japan. We observed efficacy in both AML and ovarian cancer. Notably, in AML, 4 out of 14 patients achieved complete remission—meaning the cancer temporarily disappeared from their bodies—which we consider a strong outcome. We believe these results are on par with other single-agent therapies recently approved by the U.S. FDA for AML.
We are currently conducting Phase 1/2 clinical trials in the U.S., the world’s largest pharmaceutical market. Concurrent with today’s financial results announcement, we issued a press release, and we have also published the Japanese trial results for hematological tumors in a scientific paper.
Development Milestones for rogocekib – Steady Progress with 96 Patient Enrollments Across Japan and the U.S.

We will outline the development milestones for rogocekib. In the fiscal year ended August 2025, we achieved three major milestones.
First, we secured approval to use rogocekib as the international nonproprietary name for CTX-712. Second, we obtained orphan drug designation from the FDA. Third, we completed dosing for 36 patients in the U.S., bringing the total number of patients treated in Japan and the U.S. to 96.
Looking ahead, we plan to announce interim results from our ongoing U.S. trial around mid-2026. We aim to start the Phase 2 clinical trial by mid-2027 and submit our marketing authorization application by the end of 2028.
These milestones reflect a strategic adjustment to our original timeline in response to the FDA’s Project Optimus. We recognize that addressing Project Optimus is essential for U.S. regulatory approval, and we view this as a strategic decision aimed at maximizing the value of rogocekib.
INN for CTX-712 is rogocekib

Let me explain the origin of the name “rogocekib.” The initial “r” comes from the “R” in RNA, and the “c” in the middle represents the “C” in CLK. In addition, the “o”s in the middle are treated as introns, with the entire word “rogocekib” designed to evoke the image of an RNA splicing reaction. We hope you will remember the name rogocekib.
Received Orphan Drug Designation in U.S.

I will explain Orphan Drug Designation (ODD). ODD refers to the designation for drugs intended to treat rare diseases. This designation provided several benefits, including exemptions from application fees, tax benefits, and data protection for up to seven years after approval.
We do not regard this designation merely as a means of obtaining preferential treatment. Rather, we believe that it exemplifies how we have demonstrated the value of rogocekib through close communication with regulatory authorities, particularly the U.S. FDA.
Our Approach to FDA Guidance Project Optimus

The FDA’s Project Optimus provides guidance for optimizing oncology drug dosing from the early stages of clinical trials. We are evaluating a twice-weekly dosing schedule, in addition to a once-weekly schedule. We also plan to increase the size of the expansion cohort following dose escalation beyond what was originally planned, to between 60 and 70 patients, to compare multiple dosing regimens.
This approach will temporarily extend the development timeline but reduces the risk that the application for approval will be rejected. We can also enhance the drug’s clinical value by thoroughly evaluating its efficacy and safety in clinical trials. This provides a competitive advantage in pricing negotiations and market penetration after approval. Our response to Project Optimus is based on a strategic decision to maximize the value of rogocekib.
CDK12 Inhibitor CTX-439
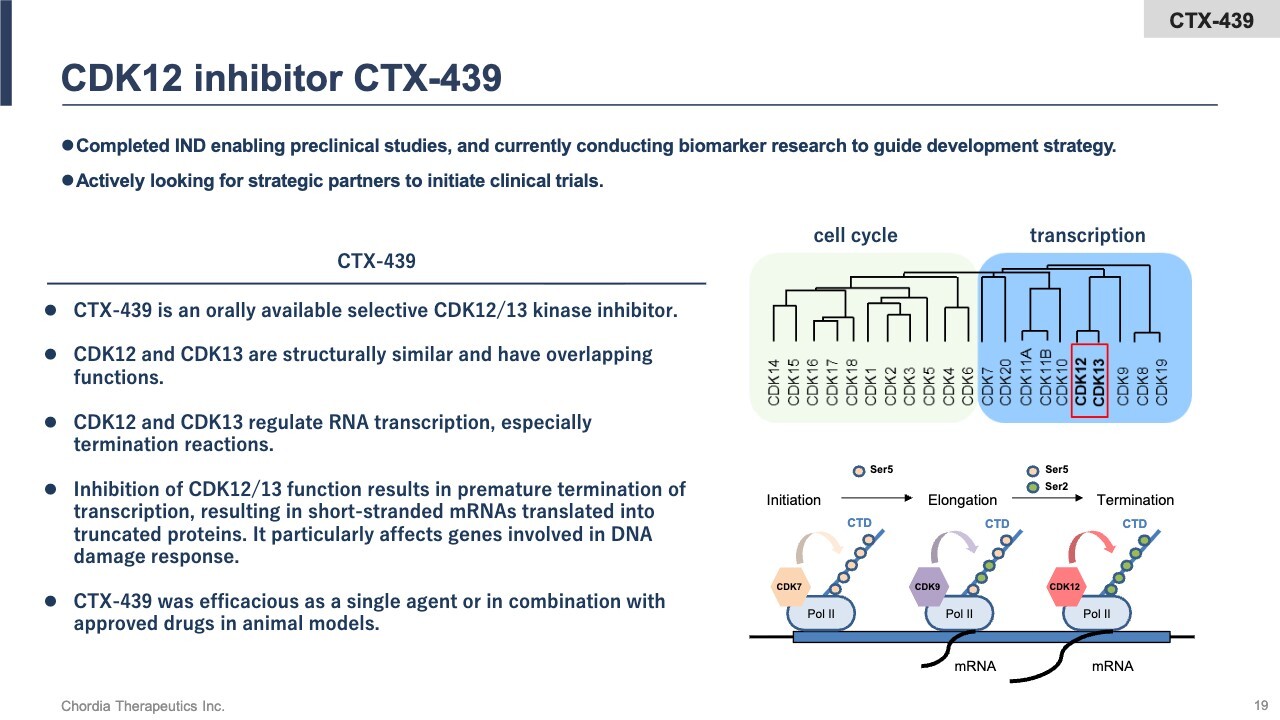
Next, I will discuss our third and fourth pipeline programs: the CDK12 inhibitor and the GCN2 inhibitor, which comprise our RNA stress response pipeline.
For the CDK12 inhibitor, we have completed preclinical data collection on a compound called CTX-439. CDK12 is a kinase that primarily regulates the termination phase of RNA transcription. CTX-439 inhibits CDK12 activity and terminates transcription prematurely, leading to the generation of abnormal RNA. This triggers stress, resulting in the death of cancer cells. We are currently seeking a partner to initiate clinical trials.
GCN2 Inhibitor CRD-099
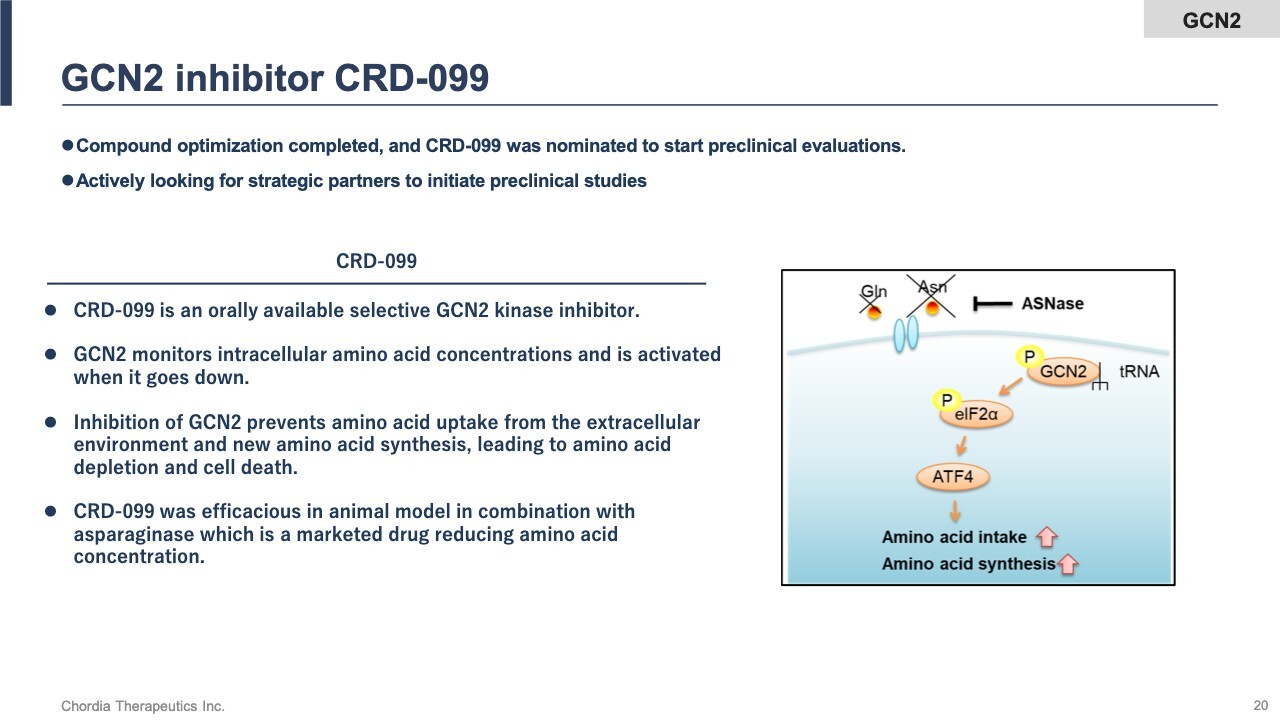
The GCN2 inhibitor is a kinase that monitors intracellular amino acid concentrations. Inhibiting its activity disrupts the translation process by which proteins are synthesized from RNA, inducing cellular stress. As a result, cancer cells are eliminated. Although this project is still in the exploratory research phase, we are seeking a partner because we are focusing our resources on rogocekib.
Joint Research with DWTI
For pipeline programs other than rogocekib, we are exploring potential applications beyond oncology through collaborative research. In partnership with D. Western Therapeutics Institute (DWTI), we are investigating the potential use of our compounds in the ophthalmology field, specifically for eye-related diseases.
Joint Research with SENJU
We are conducting joint research on the GCN2 inhibitor with Senju Pharmaceutical Co., Ltd., exploring its potential applications in ophthalmology.
In this way, while focusing on rogocekib, we are actively pursuing initiatives across multiple pipeline programs to create new value.
Four Compound Patents Supported by Multiple Use and Process Patents
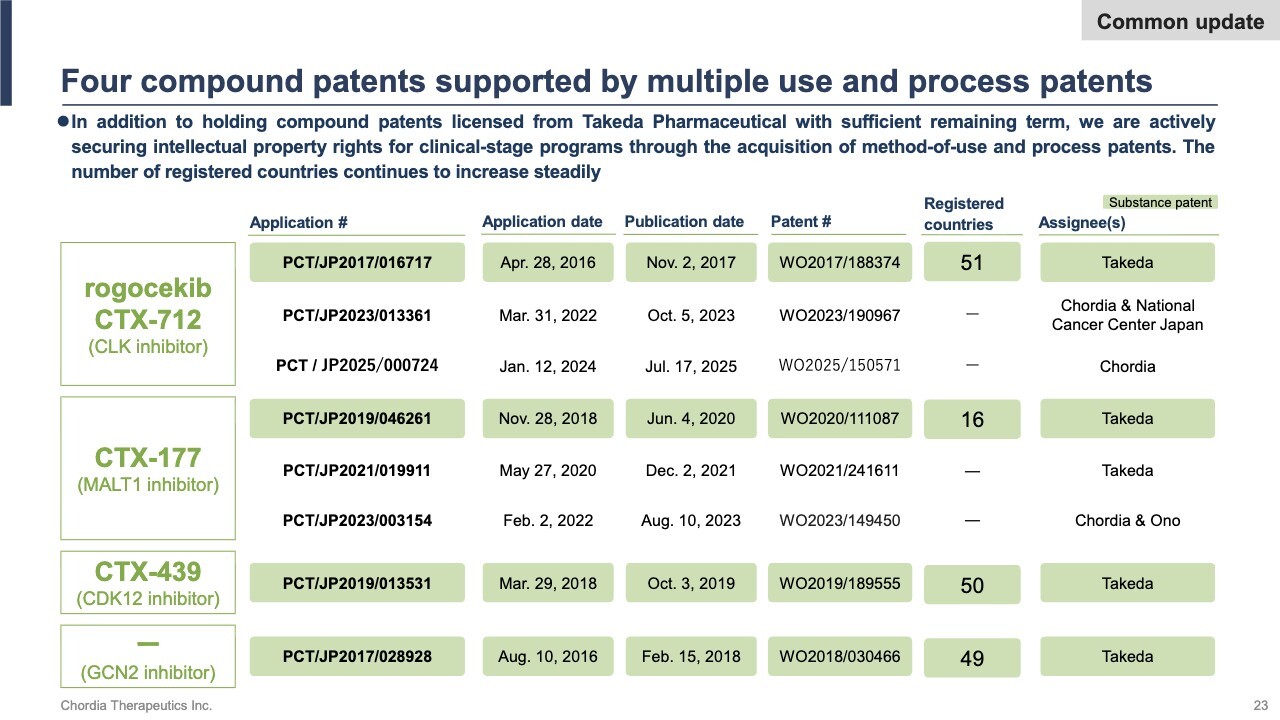
Regarding the patent status, registrations for compound patents covering rogocekib, CTX-177, CTX-439, and the GCN2 inhibitor are progressing smoothly in various countries and regions. For rogocekib and CTX-177, which are still in the clinical stage, we have filed applications for use and process patents to secure intellectual property rights.
Patents are an important factor not only in enhancing competitiveness during post-approval drug pricing and licensing negotiations, but also in securing favorable terms at the time of out-licensing. We will continue to strengthen our intellectual property portfolio to ensure a competitive edge in the global market.
Steady Progress in Both R&A and Corporate Activities
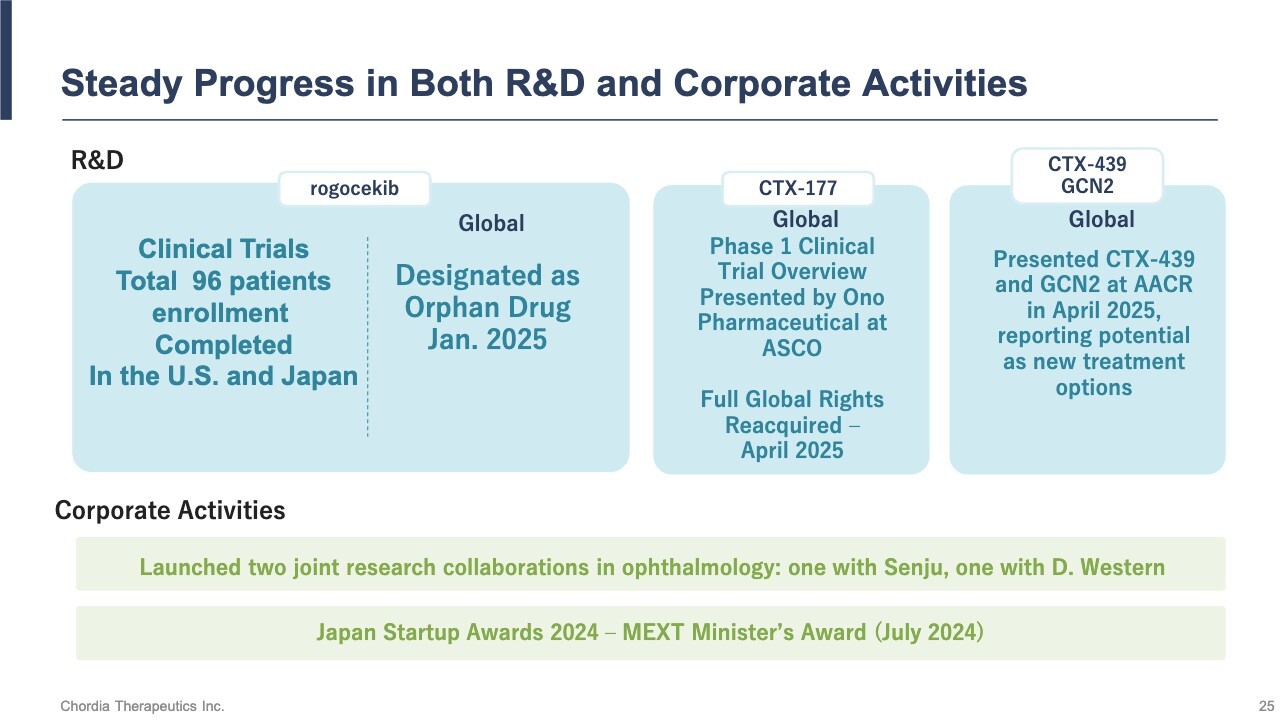
This is a review of our business and outlook. Regarding the development of rogocekib, we have revised the trial schedule in the U.S. to meet Project Optimus requirements. However, patient enrollment in the clinical trials is largely progressing as planned. As of the end of August, we had administered doses to 96 patients—36 in the U.S. and 60 in Japan. We have also received Orphan Drug Designation from the FDA.
For CTX-177, we have reacquired the rights from Ono Pharmaceutical Co., Ltd. We are actively seeking a new out-licensing partner to resume clinical trials as soon as possible.
With respect to our other preclinical pipeline programs, we have presented research findings at an international conference (the American Association for Cancer Research), and we are continuing to explore new value through collaborative research.
Prioritized Business Goals for FY25

These are our priority business goals for the fiscal year ending August 31, 2026. First, we will steadily advance the development of rogocekib. In terms of business development, we will mainly focus on CTX-177. We have received the complete set of data from the clinical trials conducted by Ono Pharmaceutical. We will actively utilize this data to negotiate the early resumption of clinical trials.
We will share updates on these developments with our shareholders and investors at appropriate times.
Management Policies and 2030 Vision

Finally, let me share our management policies. As a biotech company headquartered in Japan, we are committed to delivering innovative anti-cancer drugs that function differently from existing treatments to patients battling cancer around the world. We aim to create a society where patients and their families can look to tomorrow with hope, believing that tomorrow will bring better treatment than today.
We aim to develop new drugs and grow into a global biotech company with the support of many people. It takes a long time to bring a new drug to market, and we still need three more years before we can apply for approval. We sincerely appreciate your continued support.
Q&A Session: Status of out-licensing for CTX-177
Question: You mentioned that you are seeking an out-licensing partner for CTX-177. Could you please share the current status, within the scope of what you are able to disclose?
Miyake: Ono Pharmaceutical notified us on April 28 of its decision to discontinue the development of CTX-177 and to terminate the license agreement. The agreement has now ended, and we hold all rights to CTX-177 worldwide.
We are continuing detailed discussions on the final logistics for the transfer of all clinical and non-clinical trial data generated by Ono Pharmaceutical, as well as all CTX-177-related intellectual property rights filed by Ono Pharmaceutical, to us. At this point, the transfer of nearly all data has been completed.
Therefore, we are currently reaching out to companies around the world to ask whether they would be interested in the resumption of clinical trials for the MALT1 inhibitor CTX-177. We are working diligently as a company to find a partner as soon as possible and resume clinical trials.
Q&A Session: Purpose and details of the expansion cohort trial for rogocekib
Question: You mentioned that an expansion cohort trial for rogocekib is about to begin. I would like to follow up and ask for additional details regarding its purpose and what exactly will be involved.
Miyake: We have already completed the Phase 1 clinical trial in Japan involving 60 patients. In Japan, we used capsules for the clinical trial. We conducted the trial with a very rudimentary method of packing rogocekib into capsules, and we obtained favorable results. Based on these results, we determined that we needed to develop a better formulation for the approval application, and we subsequently manufactured a new tablet.
We are currently conducting a Phase 1 clinical trial in the U.S. using this tablet. In general, Phase 1 clinical trials are conducted in two stages: a dose escalation phase followed by an expansion phase. We are currently evaluating a 100-milligram dose administered twice weekly as the final step of the dose escalation phase. We plan to complete enrollment this year and initiate the expansion phase once the evaluation of dose-limiting toxicity (DLT) is complete.
In the expansion phase, clinical trials have typically been conducted using the maximum tolerated dose permitted in the dose escalation phase. However, we intend to proceed in accordance with the FDA’s Project Optimus and follow its guidance to compare and evaluate multiple dosing regimens. Based on the results of the dose escalation phase, we plan to select two or three dosing regimens and conduct a comparative evaluation in stages.
As a result, the planned enrollment for the expansion phase will increase to between 60 and 70 patients. We believe that, by carefully comparing dosing regimens and choosing the optimal option before starting the Phase 2 clinical trial, we can improve the chances of its success.
